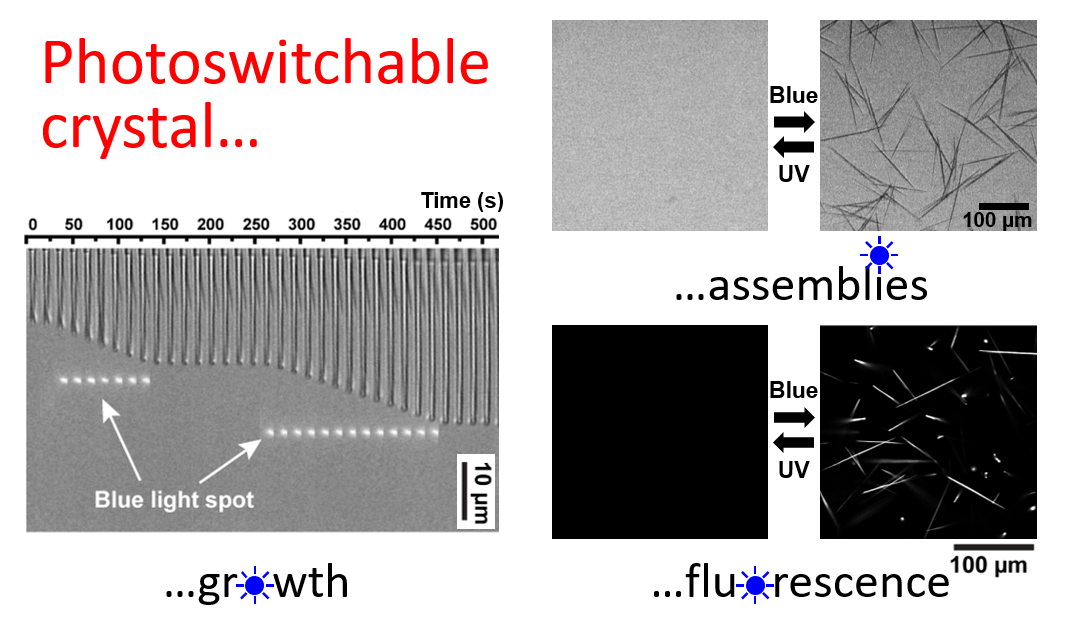
Abstract: Self-assembled nucleobases, such as G-quartets or quadruplexes, have numerous applications but light-responsive structures are limited to small, non-crystalline motifs. In addition, the assembly of the widely exploited azobenzene photochromic compounds can produce fluorescent crystals of extended dimensions but at the prize of sacrificing their photoswitchability. Here we overcome inherent limitations of self-assembly with a new concept of supramolecular co-assembly leading in fine to materials with unprecedented properties. We show that the co-assembly of guanosine monophosphate (GMP) with an azobenzene-containing DNA intercalator produce supramolecular crystals arranged through a combination of π-π, electrostatic and hydrogen bond interactions. The resulting crystals are 100 µm long, pH-sensitive, fluorescent and can be photoreversibly disassembled/reassembled upon UV/blue irradiation. This allows us to perform operations such as dynamic photocontrol of a single crystal growth, light-gated permeability in membrane-like materials and photoswitchable fluorescence. We believe this concept critically expands the breadth of multifunctional materials attainable by self-assembly.
Reference:
Photoswitchable fluorescent crystals obtained by the photoreversible co-assembly of a nucleobase and an azobenzene intercalator
L. Zhou, P.Retailleau, M. Morel, S.Rudiuk, D. Baigl*
J. Am. Chem. Soc. 2019, 141, 9321–9329
 -doi : 10.1021/jacs.9b02836
-doi : 10.1021/jacs.9b02836
Reference:
Photoswitchable fluorescent crystals obtained by the photoreversible co-assembly of a nucleobase and an azobenzene intercalator
L. Zhou, P.Retailleau, M. Morel, S.Rudiuk, D. Baigl*
J. Am. Chem. Soc. 2019, 141, 9321–9329